Inflammatory microenvironment-educated MSCs produce exosomes with enhanced regenerative potential in burn wounds
To screen the pro-regenerative miRNAs from MSC-Ex, we aimed to specifically enhance the reparative properties of MSC-Ex. Following this, miRNA sequencing will be applied to assess the miRNA composition and relative expression levels of each miRNA between the functionally enhanced and regular MSC-Ex. This function-oriented miRNA sequencing approach is supposed to aid in screening the key miRNAs that promote wound healing within MSC-Ex.
Previous studies have reported that MSCs can adjust the composition and functionality of their secreted exosomes in response to the distinct microenvironments in which they reside [23]. Herein, macrophages were induced by lipopolysaccharide (LPS) to undergo a transition to an inflammatory state (Figure S1). Subsequently, the culture medium containing the secreted products from these inflammatory macrophages was collected and concentrated (Fig. 1A), designated as CCM. To mimic the inflammatory microenvironment that MSCs may encounter when migrating into the wound tissue and to enhance the pro-regenerative function of their secreted exosomes, MSCs were educated with varying concentrations of CCM. It was shown that MSCs exposed to 10% and 5% CCM (designated MSC10 and MSC5) displayed abnormal morphology and suppressed proliferation (Fig. 1B, C and Fig. S2) as compared with the control MSCs (MSC0), consistent with literature findings [24, 25]. Additionally, we made the initial discovery that MSCs exposed to 1.25% CCM (named MSC1.25) exhibited enhanced proliferative capability (Fig. 1B, C). Then, MSC-Ex were prepared from the medium via ultracentrifugation. They were characterized as having a standard size distribution (30–130 nm) and exhibiting typical exosomal markers of CD9 and CD81 (Fig. 1D-F).
The induction of MSCs and the identification of exosomes. (A) Schematic diagram of MSCs induction. (B,C) Western blotting and quantitative analysis of proliferating cell nuclear antigen (PCNA, a marker of cell proliferation) in MSCs, with “Lamin b1” as the loading control. Statistical analysis was performed by one-way ANOVA with Tukey’s multiple comparison test, **P < 0.01, ns, not significant. Data are presented as mean ± S.D. (D) Size distribution of MSC-Ex. (E) Representative images of transmission electron microscopy (TEM) analysis of MSC-Ex. Scale bar: 100 nm. (F) Western blotting analysis of surface markers (CD9 and CD81) of MSC-Ex
Promoting the proliferation of skin cells, reducing inflammatory responses, and preventing fibrogenesis are common strategies for accelerating wound healing and preventing scar formation [26]. Functional assays revealed significant disparities in pro-proliferative, anti-inflammatory, and anti-fibrotic efficacy among exosomes secreted by MSCs stimulated with CCM at concentrations of 10%, 5%, 2.5%, 1.25%, and 0%, respectively (named EX10, EX5, EX2.5, EX1.25, and EX0). In the CCK-8 assay (Fig. 2A), EX1.25 exhibited significantly enhanced (139.07 ± 5.65%) activity in promoting the proliferation of mouse dermal fibroblasts compared to EX0 (118.14 ± 8.09%), while EX10 and EX5 had no significant effect on the proliferation of cells. Similarly, cells treated with EX1.25 exhibited the highest PCNA expression (Fig. 2B), indicating the greatest proliferative capacity. The in vitro anti-inflammatory assays (Fig. 2C) demonstrated that all five groups of exosomes significantly inhibited the nitric oxide (NO) synthesis in macrophages induced by LPS, when compared with the negative control (NC) group. Notably, macrophages treated with EX1.25 showed the lowest levels of NO synthesis (0.64-fold of NO expression vs. EX0 group). Western blotting analysis of nitric oxide synthase 2 (NOS2) expression (Fig. 2D) in macrophages of various groups demonstrated similar results. To further validate this conclusion, the levels of inflammatory cytokines interleukin-1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α) in the supernatants of various cell groups were also measured (Figure S3), with results consistent with those depicted in Fig. 2C and D. Upon stimulation with transforming growth factor-beta 1 (TGF-β1), dermal fibroblasts undergo polarization, leading to the substantial synthesis of α-smooth muscle actin (α-SMA) and the induction of tissue fibrosis, which results in scar formation [3, 4]. The in vitro gel contraction assay is a classical and effective method for evaluating the degree of fibroblast polarization. It was demonstrated that all groups of MSC-Ex significantly inhibited the TGF-β1-induced gel contraction capacity of fibroblasts, with the EX1.25 treated group showing the lowest rate of contraction (Fig. 2E, F). Western blotting analysis of α-SMA further indicated that EX1.25 exhibited the strongest antagonistic effect on TGF-β1-induced α-SMA synthesis (Fig. 2G). These results demonstrated that EX1.25 possess the best proliferative, anti-inflammatory, and anti-fibrotic functions among the five groups of MSC-Ex; meanwhile, the stimulus intensity of the inflammatory environment determines the ultimate functional orientation of MSC-Ex. Based on this, subsequent research will focus on EX0 and EX1.25.
The deep second-degree burn wound model was established in mice to compare the in vivo wound healing and anti-scarring activities of EX1.25 and EX0. Compared with the control group, both EX1.25 and EX0 significantly promoted burn wound healing and mitigated scar formation (Fig. 2H). Additionally, the EX1.25 group demonstrated a stronger wound healing capability than the EX0 group, along with a reduction in scarring. On day 6, the percentage of the remaining wound area in the EX1.25 group and the EX0 group was 40.16 ± 5.44% and 54.31 ± 13.14% (Fig. 2I), respectively. By day 9, these values decreased to 9.47 ± 6.39% and 23.01 ± 10.61% in the EX1.25 group
Comparison of the biological functions of EX0, EX10, EX5, EX2.5, and EX1.25. The CCK-8 assay (A) and the western blotting analysis of PCNA (B) were carried out to evaluate the pro-proliferative effect of EX0, EX10, EX5, EX2.5, and EX1.25 on dermal fibroblasts. The effects of the five groups of MSC-Ex on the LPS-induced inflammatory response were assessed. NO (evaluated by nitrite content) synthesis (C) and relative expression of NOS2 (D) in macrophages were tested, with GAPDH as the loading control. Gel contraction assay (E, F) and western blotting analysis (G) were performed to compare the anti-fibrotic effect of the five groups of MSC-Ex on TGF-β1-activated dermal fibroblasts. Higher gel contraction percentages (E) or smaller collagen matrices (F) indicate stronger fibrogenesis. For western blotting analysis (G) of α-SMA (a marker of fibrosis) in TGF-β1-treated dermal fibroblasts, the higher α-SMA expression suggests a higher fibrotic level. In vivo pro-regenerative effects of EX0 and EX1.25 were compared. (H) Photographs of representative wounds from each group on different post-wound days. Scale bar: 2 mm. (I) Quantification of the residual wounds; significance was compared with the control group. (J) Photomicrography of wounds (H&E staining). In the images of the wound tissue on Day 6, the necrotic tissue is on the left side of the blue dashed line, while the regenerated tissue is on the right side of the blue dashed line. Scale bars: 200 μm (left) or 50 μm (right). All statistical analyses were performed by one-way ANOVA with Tukey’s multiple comparison test, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. Data are presented as mean ± S.D
and the EX0 group, respectively. The results of H&E staining (Fig. 2J) revealed that on day 6, both the EX0 and EX1.25 groups showed greater granulation tissue formation at the wound sites compared to the control group, with the EX1.25 group exhibiting superior granulation tissue development than the EX0 group. In the subsequent healing process, compared to the control group, both the EX0 and EX1.25 groups showed a faster extracellular matrix formation. Furthermore, by day 18, wounds treated with EX1.25 demonstrated enhanced regeneration of cutaneous appendages, such as hair follicles and sweat glands (marked with blue arrows), compared to wounds treated with EX0. These results indicate that EX1.25 possess a superior pro-regenerative capacity for wound healing over EX0.
The miRNAs derived from MSC-Ex have significant pro-proliferative, anti-inflammatory, and anti-fibrotic abilities
To pinpoint the key miRNAs exerting pro-regenerative effects within MSC-Ex, miRNA sequencing was conducted on the miRNAs within EX0 and EX1.25. The study revealed that a total of 682 miRNAs were expressed across the two groups of exosomes (Fig. 3A and Table S1). Significant differences were observed in the relative abundance of many miRNAs between the groups (Fig. 3B, C). In comparison to EX0, EX1.25 exhibited a significant upregulation of 40 miRNAs and a significant downregulation of 77 miRNAs. Given that EX1.25 demonstrated a higher pro-regenerative capacity compared to EX0, the miRNAs that play a primary role should be more abundant in EX1.25 or exhibit high abundance in both exosome groups. Oriented by this principle, 32 miRNAs were pre-screened for further exploration (Table S2), including four novel miRNAs (2_18240, 3_21147, 5_24979, and 6_26497). The 32 miRNAs accounted for 84.05% and 79.92% (Fig. 3D) of total miRNAs in EX1.25 and EX0, respectively.
The differential expression of miRNAs between EX1.25 and EX0. (A) Differences in miRNA composition between EX1.25 and EX0. (B) The number of differentially expressed miRNAs in EX1.25 and EX0. (C) The clustering heatmap of differentially expressed miRNAs in EX1.25 and EX0. (D) Percentage distribution of the 32 miRNAs screened in EX1.25 and EX0
The study of miRNAs has emerged in recent years, and the functions of many miRNAs have not yet been well determined. Herein, functional studies were performed on the pre-screened 32 miRNAs. The CCK-8 assay showed that 15 of these miRNAs promoted the proliferation of dermal fibroblasts (Fig. 4A). This conclusion was further substantiated by western blotting analysis, with cells transfected with these 15 miRNAs exhibiting elevated levels of PCNA expression (Figure S4). To the best of our knowledge, the pro-proliferative effect of the majority of these miRNAs on skin cells was first identified, excluding miR-145a-5p, miR-21a-5p, miR-24-3p, and miR-92a-3p. The results of the anti-inflammatory assays demonstrated that the 32 miRNAs were capable of suppressing the LPS-induced inflammatory phenotype transition in macrophages to varying extents. In comparison with the NC group, the experimental groups transfected with these miRNAs displayed significantly reduced expression levels of NO (Fig. 4B) and NOS2 (Figure S5). The assessment of inflammatory cytokines IL-1β and TNF-α (Figure S6) in the supernatants of various cell groups also yielded concordant findings. Notably, 2_18240, miR-574-3p, 6_26497, miR-130a-3p, and miR-143-3p demonstrate relatively weaker anti-inflammatory capabilities compared to other miRNAs. The results from the gel contraction assay (Fig. 4C and Figure S7) indicate that all miRNAs possess a certain degree of anti-fibrotic capability, with 3_21147, let-7e-5p, and miR-125a-5p demonstrating particularly prominent (gel contraction % < 30) anti-fibrotic effects among the 32 miRNAs tested. These results were further corroborated by western blotting analysis, and the findings from both assays were largely consistent (Figure S8). The anti-inflammatory and anti-fibrotic functions of several miRNAs (2_18240, 3_21147, 5_24979, 6_26497, miR-5126, let-7d-3p, miR-2137, and miR-6240) were reported for the first time globally.
Effects of the 32 miRNAs on dermal fibroblast proliferation, LPS-induced inflammatory responses, and TGF-β1-driven fibrosis. (A) Results of the CCK-8 assay. (B) NO synthesis of macrophages. (C) Results of the gel contraction assay. All statistical analyses were performed by one-way ANOVA with Tukey’s multiple comparison test. For (A), significance was compared with the control group; for (B) and (C), significance was compared with the NC group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. Data are presented as mean ± S.D
Given the results presented above, four miRNAs (2_18240, miR-574-3p, 6_26497, and miR-143-3p) will be excluded from subsequent studies. These miRNAs demonstrated slight anti-inflammatory capabilities and unimpressive anti-fibrotic effects; furthermore, 2_18240 and miR-143-3p specifically lacked the capacity to enhance the proliferation of dermal fibroblasts.
To better elucidate how the final-screened 28 miRNAs exert their functions in promoting proliferation, anti-inflammation, and anti-fibrogenesis, in silico analysis was conducted to identify the targets of these miRNAs. The signaling pathways commonly targeted for macrophage-directed therapies in clinical applications have been well defined [27]. The results of in silico analysis suggested that the 28 miRNAs can downregulate the expression of most signaling proteins within these pathways (Figure S9), such as the TLR, TNF, and IFN pathways, thereby mitigating inflammatory responses. Similarly, the classic fibrosis-associated signaling pathways, such as the TGF-β1/SMAD and the Wnt/β-Catenin pathways, are intensively regulated by these miRNAs (Figure S10). miRNAs with pro-proliferative functions typically exert their effects by downregulating the expression of proteins that possess anti-proliferative or pro-apoptotic activities [28, 29]. Several miRNAs are supposed to target the signaling proteins that have an inhibitory effect on cell proliferation, such as the p53/p21 signaling pathway (Figure S10). It should be noted that only well-recognized signaling pathways are depicted in Figures S9 and S10, with many other less-established related signaling pathways not being listed. Consequently, some miRNAs that are not shown may exert their regulatory effects through other signaling pathways.
The healing process of wounds is extremely complex, and the understanding of this process is still far from adequate. As is currently known, the repair process involves multiple signaling pathways [30, 31], Given the intricacies of dermal architecture and the inherent reparative mechanisms of wound healing, the modulatory capacity of individual bioactive constituents is typically confined to a limited array of signal transduction pathways and cellular behaviors [32, 33]. The extensive regulatory influence exerted by these 28 miRNAs on the wound healing process underscores their formidable potential for therapeutic exploitation. This suggests that the combined action of these miRNAs may offer advantages over traditional therapeutics.
The synthetic exosome-like vesicles display similar characteristics to natural MSC-Ex
miRNAs are vulnerable to degradation by ubiquitous ribonucleases, and free miRNAs are seldom internalized by cells [34]. Therefore, it is essential to develop a nanocarrier with high encapsulation efficiency for miRNAs and excellent resistance to RNase degradation to deliver miRNAs into cells. Furthermore, after the cells have engulfed the nanocarriers loaded with miRNAs, it is crucial for the miRNAs to be successfully released from the lysosomes, enter the cytoplasm, bind to mRNAs, and thus exert their regulatory effects on cellular behavior.
Previous studies have shown that exosomes can be effectively internalized by cells, with a high lysosomal escape rate for the loaded cargos [35, 36]. To ensure efficient cellular uptake and a high lysosomal escape rate of miRNAs, developing a novel nanocarrier that mimics natural exosomes is a promising direction. In recent years, plant-derived extracellular vesicles have been demonstrated as a novel and promising type of nanoscale drug delivery vehicle. They possess structural features similar to those of animal-derived exosomes, exhibit good biocompatibility, and are more cost-effective and have a significantly higher production yield compared to animal-derived exosomes [37, 38]. Watermelon, which is not only inexpensive and readily available but also has extracts that have been proven to possess certain anti-inflammatory and wound healing properties [39, 40], can serve as an ideal source of plant-derived extracellular vesicles. Herein, extracellular vesicles derived from watermelon were isolated and designated as wEV. The average hydrated particle size of wEV was shown to be 55.27 nm, and it was demonstrated that they carry a negative charge (Fig. 5A, B). In our experiment, 23.87 ± 2.63 mg of wEV (mass of protein) were obtained per liter of watermelon juice. In vitro assays demonstrated that wEV significantly enhanced the proliferation of dermal fibroblasts (Fig. 5C-E), exhibiting a clear concentration-dependent effect. Furthermore, wEV exhibited excellent immunocompatibility and did not induce macrophages to polarize towards an inflammatory state (Fig. 5F-H and Figure S11). On the contrary, they can inhibit the inflammatory polarization of macrophages triggered by LPS. Next, we found that wEV did not induce fibroblasts to transition towards a pro-fibrotic phenotype and exhibited no significant promoting or inhibitory effects on fibrosis induced by TGF-β1 (Figure S12).
Characterization, biological functions, and metabolomics analysis of wEV. (A) Hydrated particle size and zeta potential of wEV. (B) Representative images of TEM analysis of wEV. (C-E) The effects of wEV on dermal fibroblast proliferation. (C) Results of the CCK-8 assay. (D, E) Western blotting and quantification of PCNA in dermal fibroblasts treated with various concentrations of wEV. (F-H) Effects of wEV on LPS-induced inflammation and whether wEV induces the transition of macrophages to an inflammatory state. (F) Concentration of NO (evaluated by nitrite content) in the culture medium of macrophages. (G, H) Western blotting and quantification of NOS2 expression in macrophages after treatment with LPS. (I, J) Metabolomics analysis results of wEV. (I) Number of various small molecule substances in wEV. (J) Percentage content of various small molecule substances in wEV. All statistical analyses were performed by one-way ANOVA with Tukey’s multiple comparison test. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. Data are presented as mean ± S.D. For (F) and (H), significance was compared with the NC group
To clarify the material basis underlying the functions of wEV in promoting proliferation and exerting anti-inflammatory effects, a metabolomics analysis was performed. The results showed that wEV contain a total of 2,991 substances (Table S3). Among these substances, there are 683 amino acids and derivatives, 112 nucleotides and derivatives, 377 terpenoids, 331 lipids, 327 alkaloids, 228 flavonoids, 197 phenolic acids, and 124 organic acids (Fig. 5I). In addition, wEV also contain other bioactive substances, such as lignans and coumarins. Among these substances, amino acids and derivatives have the highest relative content, accounting for 37.69% of the total substances (Fig. 5J). Alkaloids, lipids, and nucleotides and derivatives account for 16.55%, 13.26%, and 8.4% of the total substances, respectively. The proportions of other substances are relatively lower. Further analysis revealed that wEV contain 14 types of coding amino acids and various amino acid derivatives (which can be processed into amino acids after entering cells), providing raw materials for protein synthesis. In addition, wEV contain various deoxynucleosides and nucleosides, which serve as direct raw materials for the synthesis of DNA and RNA. They also contain cAMP, cGMP, and tetrahydrofolate, which can indirectly promote the synthesis of DNA and RNA. Moreover, lysophosphatidylcholine, lysophosphatidylethanolamine, sphingosine, ceramide, oleic acid, linoleic acid, and their derivatives in wEV play important roles in the formation, maintenance, and functioning of cell membranes [41]. Based on these findings, we speculate that wEV may promote cell proliferation by facilitating the generation of cellular DNA, RNA, proteins, and membrane structures. As for the anti-inflammatory effects, wEV contain a variety of bioactive substances such as alkaloids, terpenoids, and flavonoids, which possess anti-inflammatory properties. This may serve as the material basis for its anti-inflammatory effects.
The above results indicate that wEV can not only be regarded as a very promising drug delivery vehicle, but its inherent “supply effect” on cell proliferation and its role in inflammation regulation are also highly beneficial for wound healing. However, current methodologies for the encapsulation of drugs within plant-derived extracellular vesicles have yielded unsatisfactory encapsulation efficiencies (such as electroporation, extrusion, and ultrasonication), particularly for large molecular-weight therapeutics [42]. To address this issue, we attempted to fuse liposomes with wEV for the development of hybrid liposome-wEV nanoplatforms to load miRNA. In recent years, researchers have found that these hybrid carriers, obtained by fusing liposomes with animal/plant-derived vesicles, not only retain the advantages of both nanocarriers but also avoid their respective shortcomings [43]. Herein, liposomes (LP) formulated with DC-Cholesterol and Dioleoyl Phosphatidylethanolamine were prepared, and subsequently loaded with miRNA mimics, yielding LP-miR. Then, LP-miR was hybridized with wEV, resulting in exosome-like vesicles (LPEx), with particle sizes and zeta potentials that closely resemble those of natural exosomes. Both MSC-Ex and LPEx exhibited hydrated particle sizes around 130 nm (Fig. 6A) with a relatively narrow distribution range [44, 45]; additionally, their zeta potentials were closely aligned. The hydrated particle size of LP was measured at 160.97 nm, and they carried a substantial positive charge. The particle sizes of these nanocarriers were further confirmed by TEM analysis (Fig. 6B).
A comparative analysis of miRNA encapsulation efficiency among LP, LPEx, and wEV revealed that both LP and LPEx exhibited high miRNA encapsulation rates, amounting to 99.15 ± 0.53% and 94.94 ± 2.58%, respectively (Fig. 6C). Conversely, wEV demonstrated a markedly low miRNA encapsulation efficiency [46], recorded at merely 6.78 ± 1.63%. Serum stability tests revealed that the unloaded LP (positively charged) were likely to adsorb the proteins (negatively charged) in exosome-free serum and form a liposome–protein corona (Figure S13), resulting in larger particles (1121.00 nm) and acquiring a negative charge [47, 48]. In contrast, the particle size of miRNA-loaded LP (LP-miR) increased slightly upon serum addition. miRNAs adsorbed on the surface of LP may provide a certain degree of steric hindrance, limiting the adsorption of LP to proteins in serum. LPEx and wEV, both of which are negatively charged, exhibited high stability in serum. Their particle size remained largely unchanged. However, a slight decrease in zeta potential was observed. This suggests that LPEx and wEV may not undergo significant adsorption in serum, indicating excellent serum
Characterization and cytotoxicity assessment of LPEx. (A) Hydrated particle sizes and zeta potential of the obtained nanovesicles. (B) Representative images of TEM analysis of the nanovesicles. Scale bar: 100 nm. (C) Comparison of LP, LPEx, and wEV encapsulation efficiency for miRNA. (D, E) The CCK-8 assay performed on dermal fibroblasts (D) and macrophages (E), respectively. (F, G) Quantitative analysis (F) and representative images (G) of live/dead staining on dermal fibroblasts. Live cells were labeled by Calcein-AM (green), and dead cells were labeled by PI (red). Scale bar: 100 μm. All statistical analyses were performed using one-way ANOVA with Tukey’s multiple comparison test. Specifically, for (D) and (E), significance was compared with the control group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. All data are presented as mean ± S.D
stability. Meanwhile, the particle size and zeta potential of LPEx and wEV treated with trypsin significantly decreased, indicating that the surface proteins of these nanovesicles may have been degraded. This further suggests that LPEx and wEV exhibit high stability in serum, with serum enzymes only capable of causing slight degradation to their surface proteins. Following that, we investigated the resistance to RNase degradation of miRNA encapsulated by LPEx. Results show that free miRNA was completely degraded when exposed to RNase A, whereas miRNA encapsulated within LPEx showed non-significant degradation (Figure S14).
Cytotoxicity is one of the non-negligible aspects of nanomedicine. Both CCK-8 assay and live/dead cell fluorescence staining results indicate that LP generated significant cytotoxicity on dermal fibroblasts and macrophages (Fig. 6D-G and Figure S15). The positively charged surface of liposomes can disrupt the cell membrane, resulting in cytotoxicity [49], which explains why LP exhibited significant cytotoxicity to dermal fibroblasts and macrophages. In contrast, LPEx and wEV exerted a significant enhancement effect on dermal fibroblast proliferation (Fig. 6D), the results of live/dead cell fluorescence staining also indicated that LPEx and wEV did not lead to cell death (Fig. 6F, G). Notably, wEV but not LPEx demonstrated a slight attenuating effect on cell proliferation to macrophages (Fig. 6E), while no significant elevated cell death was found (Figure S15). This could be one of the mechanisms through which wEV exert their anti-inflammatory function.
miRNAs encapsulated in LPEx efficiently perform their biological functions
To assess the delivery efficiency of miRNAs using LPEx, the cellular uptake kinetics of LP, LPEx, and wEV were compared. The results (Figure S16) showed that the uptake of LP, LPEx, and wEV by cells increased with the incubation time. However, for both dermal fibroblasts and macrophages, the uptake of LPEx was higher than that of LP, while the lowest uptake was observed for wEV, which may be attributed to their low loading efficiency of miRNA. These findings suggest that LPEx may be more advantageous than LP or wEV for delivering miRNA. Moreover, the lysosomal escape capabilities of miRNA delivered via LP, LPEx, and wEV were compared. The findings revealed that miRNA encapsulated by LP exhibited a low rate of lysosomal escape following cellular uptake (Fig. 7A, B) [50, 51]. In contrast, miRNA delivered by LPEx and wEV demonstrated significantly higher rates of lysosomal escape than miRNA delivered via LP upon entering the cells. Furthermore, we investigated the lysosomal escape kinetics of miRNA delivered by LPEx. It was found that in dermal fibroblasts, the lysosomal escape rate of miRNA was relatively low at 6 h post-cellular uptake, but it increased rapidly and reached its peak at 12 h (Figure S17). In macrophages, the lysosomal escape rate of miRNA delivered by LPEx was slower, reaching its maximum at 24 h post-cellular uptake.
In order to elucidate the mechanism of cellular uptake of LPEx and why LPEx exhibit high lysosomal escape properties, specific receptor-mediated clathrin-dependent internalization, micropinocytosis, energy-dependent endocytosis, and caveolin-dependent internalization were blocked by using chlorpromazine (CPZ), amiloride, NaN3, and nystatin, respectively [52]. We found that CPZ and nystatin significantly inhibited cellular uptake of LPEx, whereas amiloride and NaN3 had no significant effect on LPEx entry (Fig. 7C, D). It suggests that cells mainly rely on specific receptor-mediated clathrin-dependent internalization and caveolin-dependent internalization for the uptake of LPEx. Both types of internalization depend on the interaction between ligands on the surface of the vesicles and receptors on the cell surface. Previous studies have shown that the key reason mammalian exosomes have a high ability to escape from lysosomes is due to the interaction between exosomal surface ligands and cell surface receptors [53]. Plant-derived extracellular vesicles share similar structures and compositions with animal-derived exosomes [37, 38], which may account for their high lysosomal escape rates. However, due to the current limited research on plant extracellular vesicles, it is not yet clear which receptors on the surface of wEV are responsible. LPEx were obtained by the fusion of LP and wEV, and their surface has ligands derived from wEV. These ligands enable LPEx to have endocytic pathways similar to exosomes, which may be the reason for their high lysosomal escape ability.
Cellular uptake and miRNA delivery efficiency of LPEx. (A) Representative images of the co-localization of lysosomes (green) with Cy3-miRNA (red). Scale bar: 20 μm. (B) Quantitative analysis of lysosomal escape efficiency. (C) Representative images of the cellular uptake of LPEx on dermal fibroblasts and macrophages receiving various inhibitor treatments. Nuclei were labeled with DAPI (blue), and LPEx was labeled red. Scale bar: 20 μm. (D) Quantitative analysis of the mean cellular uptake of LPEx in each group. (E, F) Effects of the 28 miRNAs encapsulated in different nanocarriers on dermal fibroblast proliferation. (G, H) Effects of the 28 miRNAs encapsulated in different nanocarriers on LPS-induced inflammatory responses. (I-K) Anti-fibrotic effect of the 28 miRNAs encapsulated in different nanocarriers on TGF-β1-activated dermal fibroblasts. All statistical analyses were performed using one-way ANOVA with Tukey’s multiple comparison test. Specifically, for (D), significance was compared with the control group. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. All data are presented as mean ± S.D
Subsequently, to evaluate whether LPEx-delivered miRNAs can fully manifest their biological functions, LPEx, LP, and wEV were loaded with the screened 28 miRNAs to obtain LPEx-R, LP-R, and wEV-R, respectively. The percentage content of each miRNA among these 28 miRNAs is equivalent to their natural proportion in EX1.25 (Table S4). The pro-proliferative, anti-inflammatory, and anti-fibrotic efficacies of LPEx-R, LP-R, and wEV-R were then comparatively assessed against those of EX1.25 and miR-NC-loaded LPEx (LPExNC). The results indicated that both EX1.25 and LPEx-R markedly enhanced dermal fibroblast proliferation (Fig. 7E, F). In contrast, LP-R and LPExNC did not significantly influence cell proliferation. In anti-inflammatory assays, all nanovesicle groups, with the exception of LPExNC, exhibited significant anti-inflammatory effects (Fig. 7G, H and Figure S18). Notably, LPEx-R surpassed EX1.25 in efficacy, demonstrating the most potent anti-inflammatory activity (0.40-fold of NO expression vs. EX1.25 group). Anti-fibrotic experiments revealed that EX1.25 and LPEx-R both significantly reduced fibrogenesis, exhibiting comparable inhibitory effects (Fig. 7I-K). The remaining three groups of vesicles showed either no effect or only a mild inhibitory effect on fibrogenesis. These results indicate that miRNAs delivered via LPEx can efficiently enter cells and fully exert their regulatory functions on cellular behavior.
LPEx-R significantly enhance burn wound healing and reduce scar formation
Prior to assessing the in vivo efficacy of LPEx in promoting wound healing and preventing scarring, their biocompatibility was evaluated. The subcutaneous tissues of mice injected with LP, LPEx, and wEV showed no apparent necrosis and no expression of IL-1β and TNF-α. In contrast, the subcutaneous tissue of mice injected with egg white exhibited significant necrotic areas (Figure S19A) and expressed high levels of IL-1β and TNF-α (Figure S19B). In another test, hematological examinations of mice receiving LP, LPEx, and wEV treatment (intravenous injection) showed normal values for each indicator. However, mice injected with LPS exhibited abnormal ratios of lymphocytes, monocytes, and eosinophils, as well as abnormal lymphocyte and platelet counts in the blood (Table S5). Additionally, no significant upregulation in the expression levels of IL-1β and TNF-α was observed in the serum of mice injected with LP, LPEx, or wEV (Figure S20).
To investigate the organ distribution of LP, LPEx, and wEV after entering the body, these nanovesicles were intravenously injected into mice, respectively. It was found that 48 h later, LPEx and wEV were distributed in the brain, lungs, liver, spleen, and kidneys of the mice, but were mainly concentrated in the liver and kidneys (Figure S21). In contrast, LP were only detected in the liver and kidneys. Moreover, the total fluorescence intensity of LP was significantly lower than that of the LPEx and wEV groups. This may be attributed to the adsorption of LP with proteins in the blood, forming larger particles (as shown in Figure S13), which are subsequently cleared by immune cells. These results indicate that LPEx and wEV are more stable in vivo and more capable of withstanding long-circulation compared to LP. Given that LP, LPEx, and wEV are primarily distributed in the liver and kidneys, their potential impact on hepatic and renal function was evaluated. The mice received intravenous injection of normal saline (control), LP, LPEx, and wEV showed normal serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), blood urea nitrogen (BUN), and creatinine (CR) levels (Figure S22A, B). However, in mice treated with carbon tetrachloride (CCl4), there was a significant elevation in the levels of serum ALT and AST. Likewise, mice subjected to cisplatin (DDP) treatment exhibited a notable increase in BUN and CR levels in their serum, while other groups did not show such an increase. The result of H&E staining further confirms that LP, LPEx, and wEV are harmless to the liver and kidneys. Conversely, CCl4 and DDP caused significant damage to the liver (necrosis and ballooning degeneration.) and kidney (tubular degeneration, necrosis, and inflammatory cell infiltration), respectively (Figure S22C, D).
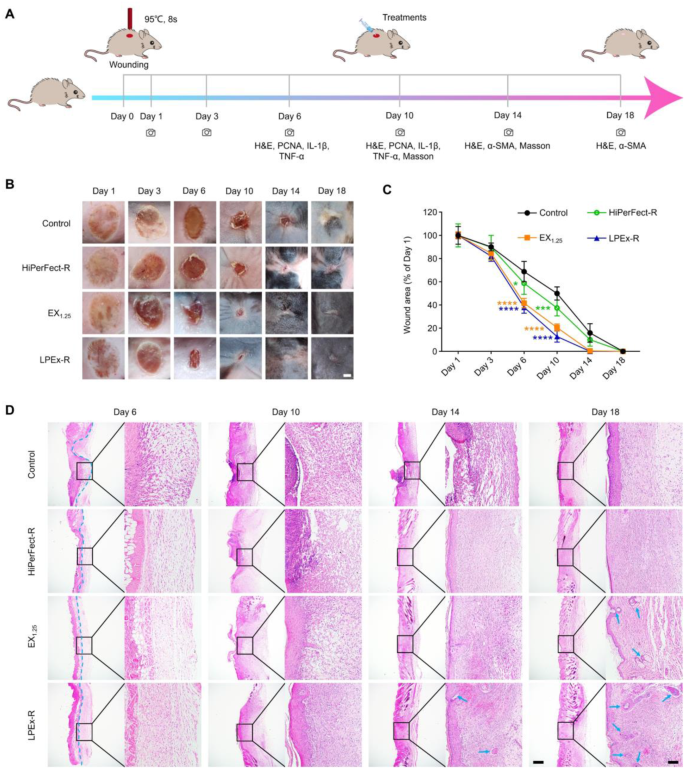
Finally, the impact of LPEx-R on deep second-degree burn wound healing and scar formation was evaluated (Fig. 8A) and compared with that of EX1.25 and the HiPerFect-R (the HiPerFect transfection reagent loaded with the 28 miRNAs). The results demonstrated that, compared to the control group, both EX1.25 and LPEx-R significantly promoted wound repair and reduced scar formation (Fig. 8B, C), while HiPerFect-R showed only a slight advantage over the control. Meanwhile, the wound healing rate in the EX1.25 group and the LPEx-R group showed no significant difference.
Comparative study of the pro-healing and scar-preventive properties of EX1.25 and LPEx-R on burn wounds. (A) Schematic diagram of the in vivo burn wound healing experiment. (B) Photographs of representative wounds from each group on different post-wound days. Scale bar: 2 mm. (C) Quantification of the residual wounds; significance was compared with the control group. (D) H&E staining of wound tissues. In the images of the wound tissue on Day 6, the necrotic tissue is on the left side of the blue dashed line, while the regenerated tissue is on the right side of the blue dashed line. Scale bar: 200 μm–50 μm. Statistical analysis was performed by one-way ANOVA with Tukey’s multiple comparison test, *P < 0.05, ***P < 0.001, ****P < 0.0001. Data are presented as mean ± S.D
Next, the microscopic structure of the wounds in each group was examined by H&E staining and Masson’s staining. The results of H&E staining (Fig. 8D) indicated that by Day 6, the control group exhibited minimal formation of granulation tissue, whereas the wounds in other groups had already developed some granulation tissue. Subsequently, on Day 10 and Day 14, the wounds in the EX1.25 and LPEx-R groups exhibited accelerated extracellular matrix synthesis as compared with the control and HiPerFect-R groups. By Day 18, the LPEx-R group displayed enhanced extracellular matrix remodeling and regeneration of skin appendages (marked with blue arrows) as compared to the EX1.25 group. In contrast, the wound tissues in the control group and the HiPerFect-R group exhibited poor remodeling of the extracellular matrix.
Collagen plays a crucial role in wound repair. It provides structural support and a scaffold for cell attachment in the wound tissue, promotes cell migration, proliferation, and differentiation, and regulates inflammatory responses and granulation tissue formation [54]. As shown in Fig. 9A and B, both LPEx-R and EX1.25 significantly promoted collagen synthesis in the wound healing process, outperforming the control and HiPerFect-R. On Day 14, the LPEx-R group exhibited the most densely and orderly arranged collagen fibers in the wound area, indicating the highest degree of remodeling. This was followed by the EX1.25 group. Although the HiPerFect-R group demonstrated a higher amount of collagen compared to the control group, the collagen was predominantly distributed in a parallel manner, which is characteristic of scar tissue [55], suggesting insufficient collagen remodeling in this group. As for the control group, the collagen content remained relatively low even by Day 14, indicating a slower rate of wound healing.
The content of PCNA can reflect the proliferative activity of cells in wound tissue. A high level of PCNA indicates that the wound is in an active repair phase, with vigorous cell division and proliferation, which promotes tissue regeneration and repair [56]. During the wound healing process, the expression level of PCNA undergoes dynamic changes. In the early and middle stages, the content of PCNA gradually increases to facilitate rapid wound filling and tissue regeneration. In the later stage, the expression of PCNA gradually decreases as the wound enters the remodeling phase, where excess cells are eliminated through apoptosis or macrophage phagocytosis, promoting wound maturation and tissue remodeling [57]. On day 6, the PCNA content
Masson’s staining and immunofluorescence staining for PCNA and α-SMA in wound tissue. (A, B) Representative images and quantitative analysis of Masson’s staining of wound tissues. Scale bar: 50 μm. (C, D) Representative images and quantitative analysis of immunofluorescence staining of PCNA (red) of wound tissues. Scale bar: 50 μm. (E, F) Representative images and quantitative analysis of immunofluorescence staining of α-SMA (red) of wound tissues. Scale bar: 50 μm. All statistical analysis was performed by one-way ANOVA with Tukey’s multiple comparison test, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns, not significant. All data are presented as mean ± S.D
in the wound tissues of the EX1.25 and LPEx-R groups was significantly higher than that in the control group (Fig. 9C, D). Although the PCNA content in the HiPerFect-R group was higher than that in the control group, it was lower than that in the EX1.25 and LPEx-R groups. These results indicate that on day 6, the wounds in the EX1.25 and LPEx-R groups were in a highly active repair phase. By day 10, the PCNA content in the wound tissues of the EX1.25 and LPEx-R groups had markedly decreased, suggesting that the cells in these wounds had exited the rapid proliferation phase and were gradually transitioning into the remodeling stage. In contrast, the PCNA content in the control group remained relatively high, indicating that the wounds in this group were still in the proliferation phase. These results further consolidated the conclusions drawn from Fig. 8B and C, demonstrating that EX1.25 and LPEx-R indeed significantly promoted wound healing.
Inflammatory responses have a direct impact on wound healing and scar formation. An appropriate inflammatory response aids in wound healing, while excessive or abnormal inflammatory responses can lead to scar formation and delayed healing. As one of the most difficult wound types to treat, burn wounds often accompany severe and persistent inflammatory responses. Therefore, modulating the inflammatory response is an important therapeutic strategy for improving burn wound healing and reducing scar formation [58]. Immunofluorescence staining for IL-1β and TNF-α was performed on the wound tissues of all groups (Figure S23), revealing that the wounds in the EX1.25 and LPEx-R groups by day 6 and day 10 exhibited significantly lower expression levels of IL-1β and TNF-α in comparison to the control group. On Day 10, the HiPerFect-R group also exhibited significantly lower levels of inflammatory cytokines in the wounds compared to the control group, yet higher than those in the wounds of the EX1.25 and LPEx-R groups. These results indicate that both EX1.25 and LPEx-R exhibited good anti-inflammatory effects in wound healing, while the in vivo anti-inflammatory effect of HiPerFect-R was inferior to that of EX1.25 and LPEx-R.
α-SMA is a marker of scarring. To further ascertain the anti-scar formation effects of HiPerFect-R, EX1.25, and LPEx-R, we conducted α-SMA immunofluorescence staining on the wound tissues. The results (Fig. 9E, F) displayed that on Day 14 and Day 18, the wounds in the EX1.25 and LPEx-R groups exhibited considerably lower expression levels of α-SMA in comparison to the control group. On Day 18, the expression of α-SMA in the wounds of the HiPerFect-R group was also lower than that in the control group. These results demonstrated that HiPerFect-R, EX1.25, and LPEx-R indeed suppressed scar formation.
The in vivo wound healing results indicate that LPEx can effectively deliver the screened 28 miRNAs to wound tissue and exerts strong pro-regenerative, anti-inflammatory, and anti-fibrotic effects, providing favorable conditions for the rapid repair of wounds and the prevention of scars. The wounds treated with LPEx-R showed similar or better outcomes compared to EX1.25 in terms of healing rate, collagen remodeling, and regeneration of skin appendages. These results provided a solid foundation for the further clinical application of LPEx-R.
One of the major concerns in the clinical translation of LPEx-R is achieving scalable production while ensuring consistent quality. In this study, the preparation processes of both LP and wEV are not complex and allow for swift large-scale production. Moreover, the fusion of LP-miR with wEV to obtain LPEx-R is a well-established process, ensuring the rapid and stable production of LPEx-R. Another concern in clinical application is the cost. LPEx-R effectively addresses this challenge by virtue of its cost-effectiveness. Watermelon is a widely available and inexpensive agricultural product, and its extracellular vesicles can be isolated using straightforward and scalable methods. Furthermore, the synthesis of LP is a robust and cost-effective process. By combining these two components, the overall production cost of LPEx-R is substantially lower than that of natural MSC-Ex. In our laboratory, the cost of preparing an equivalent quantity of LPEx-R is approximately 1/120 that of preparing MSC-Ex. This cost advantage of LPEx-R makes them more accessible for widespread clinical use.
Even though LPEx-R exhibits greater potential for widespread clinical application compared to natural MSC-Ex, there are still some unresolved issues that need to be addressed. For example, plant extracellular vesicles contain a variety of nucleic acids, such as miRNAs, which can exert a cross-kingdom effect in animal cells, regulating specific cellular functions across species [59]. However, it remains unclear whether the regulatory effects of nucleic acid components in wEV are beneficial or harmful to the organism in the context of long-term exposure. Further research is still needed to explore this question. In addition, the isolation and purification of plant extracellular vesicles still face some challenges, and a unified “gold standard” has not yet been established [60]. In the future, it will be necessary to conduct in-depth research into the biological characteristics of wEV in order to develop more efficient and specific isolation and purification methods, ensuring their safety and efficacy. Meanwhile, standardized production and quality control systems need to be established to guarantee the reliability and reproducibility of LPEx-R in clinical applications. Finally, neither wEV nor LPEx have antibacterial properties (Figure S24). However, wounds are often prone to bacterial infections due to improper or untimely treatment. Since exosomes or exosome-like vesicles are often required to be loaded into hydrogels for clinical use, we may need to develop an LPEx-R-hydrogel system in the future, endowing the hydrogel with antibacterial capabilities while achieving the stable release of LPEx-R.